
WEBINAR – Advancing Public Health with Wearables: Strategic Development of IoMT Biosensing Lifestyle Devices
On⯠April 29th, join us asâ¯MIDI Medical Device Development Corp. and Valencell, Inc. present⯠âAdvancing Public Health with Wearables: Strategic Development of IoMT Biosensing Lifestyle Devicesâ. Presented byâ¯Christopher Montalbano, CEO, MIDI Medical Device Development Corp. and Ryan Kraudel, Vice President, Marketing, Valencell, Inc., thisâ¯webinar will examine the way in which wearables advance the public health when a strategic device development approach is utilized to integrate and harmonize: Lifestyle, IoMT, Biosensing and Physiological Sensing.
Join us Tomorrow (noon EDT) for our live webinar! Register here
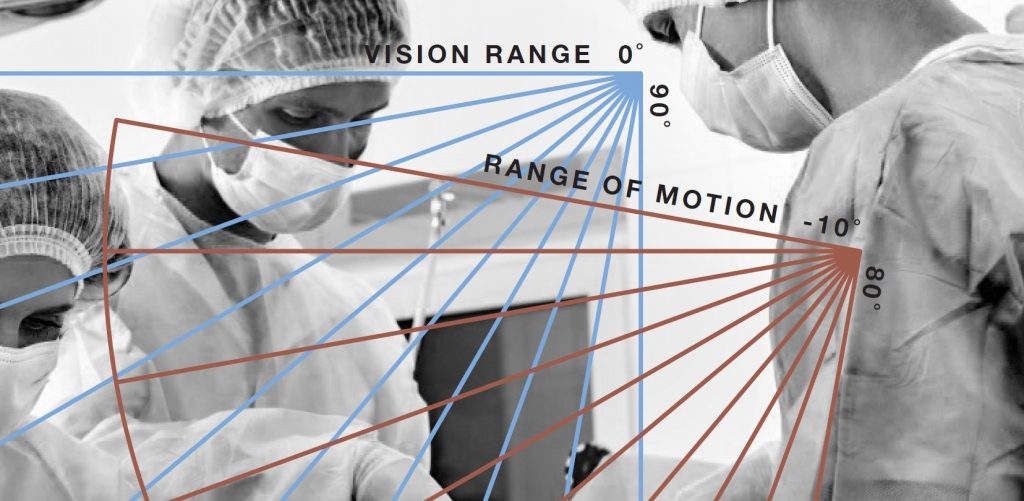
The Role of Usability Research and Human Factors in Medical Device Design, The White Paper
MIDI’s latest white paper will be avaiable next week. Stay tuned for the release of The Role of Usability Research and Human Factors in Medical Device Design. In this paper you will learn more about:
- The origin Story of Medical Device Usability Research & Human Factors Engineering
- The Fundamentals – Usability Research and Human Factors for Medical
Device Development - The Essential Baseline Components – Device Users, Use Environment and User Interfaces
- The FDA Considerations – Usability and Human Factors to Support Safe
and Effective Use - The Standards Organizations for Usability and Human Factors Considerations – AAMI, ANSI, ISO and IEC
- The First Step – Beginning your Usability and Human Factors Regulatory and Standards Roadmap
- The Top Three Main Guidance Documents – IEC, CDRH and ANSI/AAMI
- The ANSI/AAMI HE75 – Overview and Methods
- The Usability Research Methods – Generative and Evaluative Research
